Site Navigation
Oilfield expertise at your desktop
NMR Relaxation Processes
Polarization and Wait Times
The time taken to align hydrogen protons in a magnetic field is dependent on T1 time. The process of alignment is referred to as polarization. Before a CPMG experiment can be conducted, all proton spins must be aligned parallel to the B0 field. Proton spins not aligned with B0 will be tipped more than 900 when the B1 rf pulse is applied and will not lie in the x-y plane and cannot, therefore, be detected as transverse relaxation during the CPMG experiment.
The CPMG T2 experiment consists of two parts: the polarization, followed by CPMG pulse sequence. The wait time is the time that is allowed for exposure to the B0 field prior to commencement of spin tipping using the B1 rf pulse.
For proper estimation of porosity, full polarization or nearly full polarization (95%) is required. For 95% polarization, the wait time (exposure to B0 ) should be at least 3 times T1.
NMR Relaxation Processes
T2 decay is due to irreversible dephasing which occurs in three ways:
- Surface relaxation: Due to interaction of the proton spins with the surface of the pores.
- Bulk fluid relaxation:Due to molecular interactions in the fluid.
- Molecular diffusion:Due to diffusion of the molecules along magnetic gradients.
All three of these processes act in parallel and therefore, the T2 of the pore fluid is given by:1/T2 = 1/T2bulk + 1/T2 surface + 1/T2diffusion
where:
T2 = transverse relaxation
T2bulk = T2 bulk relaxation of the fluid
T2surface = T2 due to surface relaxation
T2diffusion = T2 due to diffusion processes
Surface Relaxation
In porous media, surface relaxation is due to the interaction of the proton spins in the fluid with the pore surface. The proton spins are subject to random motion, referred to as Brownian motion. When the proton spins come into contact with the pore wall, they have a finite probability of becoming irreversibly dephased by losing energy to other molecules in the pore wall.
.

T2 relaxation is related to pore size by the following equation:1/T2= rho2 (S/V)
where:
S/V = surface to volume ratio of the pore
rho2 =T2 surface relaxivity
Surface relaxivity describes the ability of the pore surface to be able to relax a proton spin. Relaxivity varies with mineralogy. Empirical data shows that for sandstones rho2 is 0.05µm/ms. Carbonates exhibit weaker relaxivity than sandstones.
The relationship between pore size (S/V) is only valid in the fast diffusion limit. The fast diffusion limit is where pores are small enough and surface relaxation slow enough that a typical molecule crosses the pore surface many times before it is relaxed.
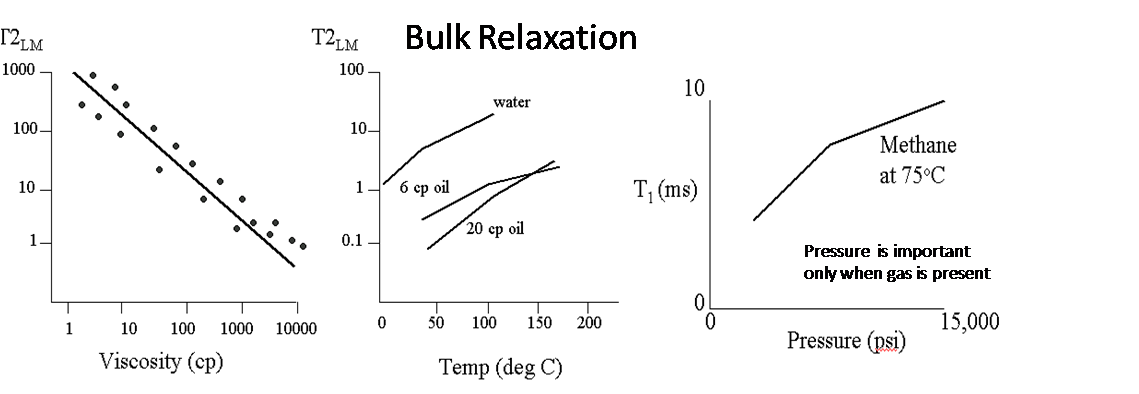
Bulk Fluid Relaxation
Every fluid has intrinsic relaxation that is determined by properties such as viscosity and chemical composition. The bulk relaxation of the fluid is not limited by surface relaxation and can only be measured by placing the fluid in a container large enough to ensure that relaxation is not considered to lie in the fast diffusion limit. T2 bulk relaxation properties also apply to the non-wetting fluid phase residing in pores (i.e., oil in water wet pores).
Molecular Diffusion
Relaxation due to diffusion is caused by molecular diffusion in the presence of a gradient field during a CPMG measurement. In the presence of a strong magnetic field, molecular diffusion causes additional dephasing and increases the T2 relaxation.
Echo Spacing
The effects of diffusion relaxation can be negated by using a short inter-echo spacing (time between CPMG pulses, TE). As the echo spacing is increased, diffusion becomes more important in the relaxation process. Gas exhibits significant diffusion even with a small inter-echo spacing (TE).
Comparing multiple CPMG experiments with variable inter-echo spacing, the diffusion coefficient can be exploited and used to identify fluids.